=================================================================================
The orbitals on adjacent atoms can be combined in two different ways to produce either a bonding or an antibonding composite orbital. Based on band theory, when a large number of atoms combine to form a large aggregate, molecular orbitals merge into an almost continuous band of energy levels. The bottom half (called valence band) of the band is composed of bonding M.O.s (molecular orbitals) and is filled with electrons. The upper half (called conduction band) of band is composed of antibonding M.O.s and is empty.
The schematic illustration in Figure 2243a shows the bonding levels for diamond. When the hybrid carbon atoms bond, a second electron is contributed to the state by the other atom, and thus the interaction between the two electrons lowers the energy of the state. Therefore, the sp3 energy level splits into a series of four bonding and four antibonding orbitals. In an actual solid, there is a Coulomb interaction between the atom cores and the electrons, and then the bonding and anti-bonding energy levels split to form a continuous band structure, with the band of bonding states being the valence band and the anti-bonding states being the conduction band.
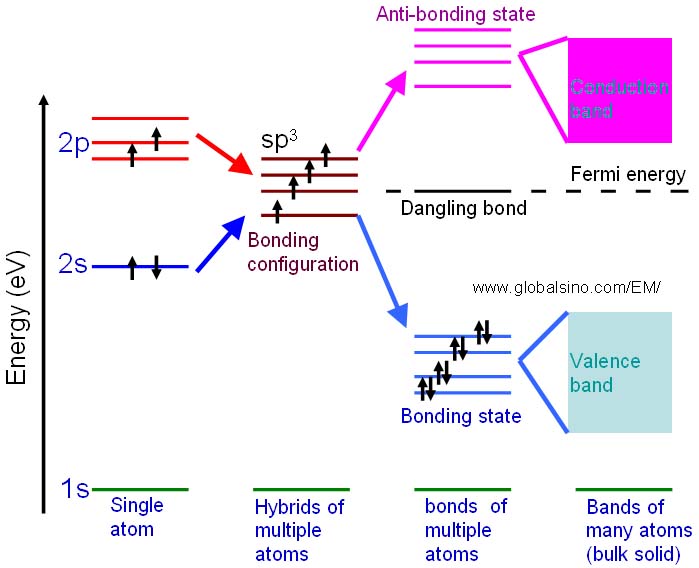
Figure 2243a. Schematic illustration showing the progression of the electronic structure for an sp3 bonded system.
However, in some materials, those electrons in different orbitals form their own energy levels of valence and conduction bands as shown in Figure 2243b.
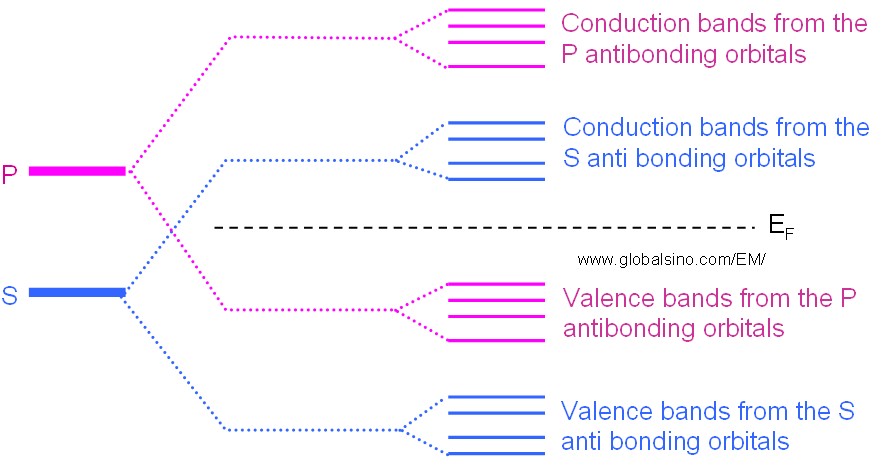
Figure 2243b. Schematic illustration of evolution of the atomic s and p orbitals into valence and conduction bands in some materials.
Figure 2243c shows the measured CeF3 F-K ELNES. [1] The main structure width, including the large peak A and its shoulder B, was evaluated as the distance between the positions of the broken lines corresponding to the minimum and maximum in the second differentiated spectra.
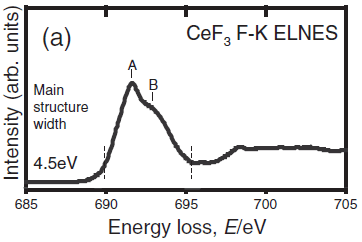
Figure 2243c. The measured CeF3 F-K ELNES. Adapted from [1]
Figure 2243d shows the unoccupied partial density of states (PDOS) of CeF3 and the bond overlap population diagram (BOPD) between Ce and its neighboring F for the non-core-holed, ground state electronic structures. The main peak structures of the ELNES in Figure 2243c correspond to the Ce5d derived states, which range from 5 to 9 eV above the Fermi level (the Fermi level in Figure 2243d is set to zero). Since the intensities of the BOPD are negative, the corresponding interactions between the Ce and anion F atoms are all antibonding. Larger BOPD intensities correspond to larger spatial overlaps between the two molecular orbitals, promoting higher antibonding energy levels. [2] The spatial overlaps between the Ce5d and the F orbitals show a variation in the Ce5d-derived states, resulting in the energy width of Ce5d PDOS and the main peak structures in the ELNES in Figure 2243c.
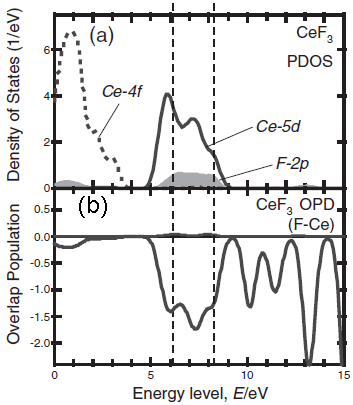
Figure 2243d. The unoccupied PDOS of CeF3 and the BOPD between Ce and its neighboring F for the non-core-holed, ground state electronic structures. Adapted from [1]
[1] Ikuo Nishida, Kazuyoshi Tatsumi and Shunsuke Muto, Local Electronic and Atomic Structure of Ce3+-Containing Fluoride/Oxide Determined by TEM-EELS and First-Principles Calculations, Materials Transactions, Vol. 50, No. 5 (2009) 952 - 958.
[2] D. G. Pettifor: Bonding and Structures of Molecules and Solids,
(Oxford University Press, 1995).
|



