Fuel Cells - Practical Electron Microscopy and Database - - An Online Book - |
||||||||||||||||||||||||||||||||||||||||||
| Microanalysis | EM Book https://www.globalsino.com/EM/ | ||||||||||||||||||||||||||||||||||||||||||
| This book (Practical Electron Microscopy and Database) is a reference for TEM and SEM students, operators, engineers, technicians, managers, and researchers. | ||||||||||||||||||||||||||||||||||||||||||
| ================================================================================= | ||||||||||||||||||||||||||||||||||||||||||
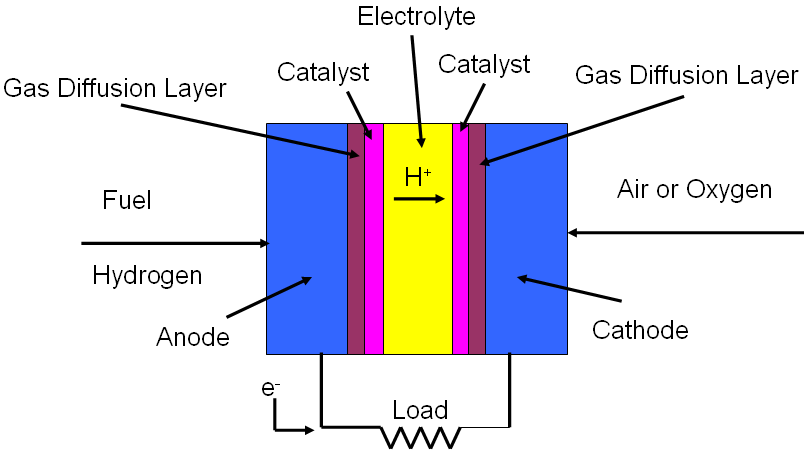
Fuel cells are devices which are used to convert chemical energy to electrical energy. As shown in Figure 3305, they normally are composed of anode, anode catalyst, cathode, cathode catalyst, gas diffusion layer, electrolyte, a load to complete the circuit, continuous feed of fuel, and continuous feed of air or oxygen. Fuel cells are generally named by the electrolytes in the cells as listed in Table 3305.
Figure 3305. Schematic illustration of a fuel cell converting chemical energy to electrical energy. Table 3305. Comparison of most common fuel cells.
|
||||||||||||||||||||||||||||||||||||||||||
| ================================================================================= | ||||||||||||||||||||||||||||||||||||||||||
| The book author (Yougui Liao) welcomes your comments, suggestions, and corrections, please click here for submission. If you let book author know once you have cited this book, the brief information of your publication will appear on the “Times Cited” page. | ||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||