=================================================================================
The misfit layer chalcogenides can have a basic formula of (AX)1+δ(BX2)n (A = rare earth, Sn, Pb, Sb, Bi; B = Ti, V, Cr, Nb, Ta; X = S, Se) [1 - 3] and can also be some minerals such as cylindrite-franckeite homologous series, legenbachite, cannizzarite [4 - 5]. δ is determined by the misfit between two sublattices and n depends on the stacking sequence. Figure 3467a shows the basic configuration of the misfit layer structures that are formed by the periodic stacking of two different types of layers (i.e. the two sublattices), i.e. one with pseudo-hexagonal symmetry labeled by H and one with pseudo-tetragonal symmetry labeled by Q. The Q-layer has edge sharing {AX5} square pyramids forming a half octahedra thick {100} slice of NaCl-type structure. The H layer can further be divided into two different configurations: when the B cation is Nb or Ta, the H layer is formed by {BX6} prisms sharing edges like in one prism thick {001} slice of molybdenite-type structure as show in Figure 3467a (b); if the B cation is Ti, V or Cr, the H layer consists of {BX6} edge-sharing octahedra like in one octahedra thick {111} slice of NaCl-type structure as shown in Figure 3467a (c). Here, each H and Q sublattice is defined in the plane of the layers by their own a and b unit cell vectors.
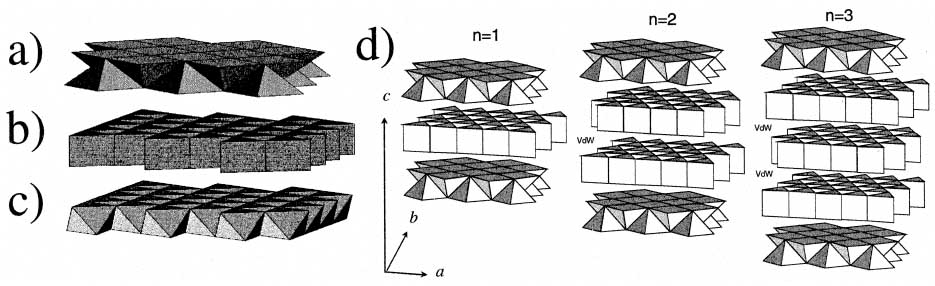
Figure 3467a. Schematic illustration of misfit layer chalcogenides. (a) The Q layer; (b) The molibdenite-type H layer; (c) The NaCl-type H layer; and (d) Stacking sequences when n = 1; n = 2 and n = 3. [6]
An example with NaCl-type H layer is La1.2CrS3.2 [7 -8] misfit layer chalcogenides with a periodical stacking of H (CrS2) and Q (LaS) layers. The repeat distance along the a axis, i.e. along the misfit direction, is different for each layer, being the coincidence unit cell 3 × aQ = 5 × aH as indicated in the electron diffraction pattern in Figure 3467b. Their mutual mismatch induces a modulation (i.e. represented by the satellite reflections), in this commensurate case, shown by the small arrows.
![Electron diffraction pattern from a crystal of LaCrS3 oriented along the [001] zone axis](image1/3467b.jpg)
Figure 3467b. Electron diffraction pattern from a crystal of LaCrS3 oriented along the [001] zone axis.
[6]
[1] Makovicky, E., Hyde, B.G., 1981. Non-commensurate (misfitt) layer structures. Struct. Bonding 46, 101 - 170.
[2] Wiegers, G.A., 1996. Misfit layer compounds. Prog. Solid State Chem. 24, 1 - 139.
[3] Wiegers, G.A., Merschaut, A., 1992. Misfit layer compounds (MS)nTS2 (M = Sn, Pb, Bi, rare earth metals, T = Nb, Ta, Ti, V, Cr; 1.08 < n < 1.23): structural and physical properties. Mater. Sci. Forum 100/101, 101 - 172.
[4] Makovicky, E., Hyde, B.G., 1981. Non-commensurate (misfit) layer
structures. Struct. Bonding 46, 101 - 170.
[5] Makovicky, E., Hyde, B.G., 1992. Incommensurate, two-layer structures
with complex crystal chemistry: minerals and related synthetics. Mater.
Sci. Forum 100/101, 1 - 100.
[6] A.R. Landa-Cánovas, A. Gómez-Herrero, L. Carlos Otero-Díaz, Electron microscopy study of incommensurate modulated structures in misfit ternary chalcogenides, Micron 32 (2001) 481 - 495.
[7] Kato, K., Kawada, I., Takahashi, T., 1977. Die Kristallstruktur von LaCrS3. Acta Cryst. B 33, 3437 - 3443.
[8] Otero-Díaz, L.C., Fitzgerald, J.D., Williams, T.B., Hyde, B.G., 1985. On the monoclinic binary-layer compound `LaCrS3'. Acta Cryst. B 41, 405 - 410.
|

![Electron diffraction pattern from a crystal of LaCrS3 oriented along the [001] zone axis](image1/3467b.jpg)