=================================================================================
Based on classical nucleation theory, the nucleation rate R can be given by,
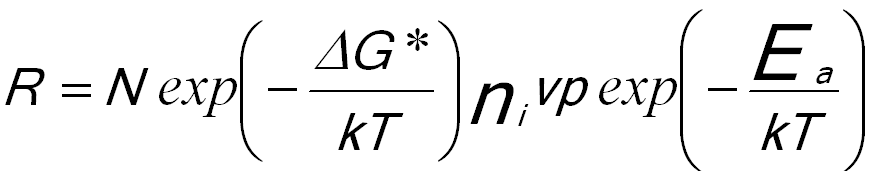 ----------------------- [1723a] ----------------------- [1723a]
= n*β ----------------------- [1723b]
where,
N -- The total number of nucleation sites per unit
volume,
ni -- The number of available atoms at
interface,
v -- The jump frequency,
k -- The Boltzmann constant,
p -- The probability
that the atomic jump will be in the proper direction,
Ea -- The activation energy for the atomic jump across the interface,
ΔG* -- The Gibbs free energy at the critical radius,
n* -- The average population of critical nuclei,
β -- The diffusion of atoms to the cluster.
The average population of critical nuclei is given by,
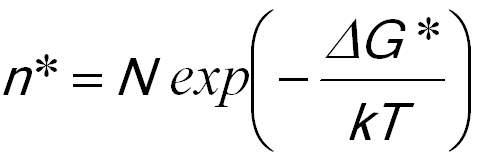 ----------------------- [1723c] ----------------------- [1723c]
The diffusion of atoms to the cluster is given by,
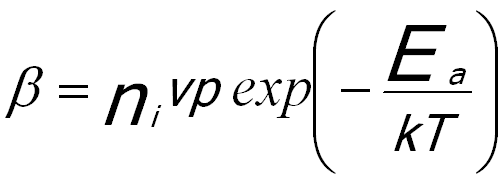 ----------------------- [1723d] ----------------------- [1723d]
Equation 1723a indicates that the nucleation rate increases exponentially as the temperature is increased.
|
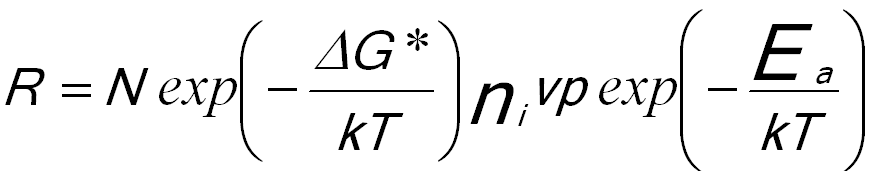 ----------------------- [1723a]
----------------------- [1723a] 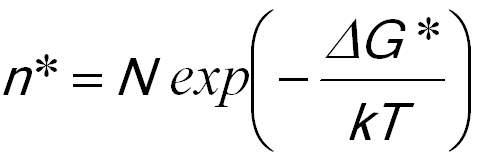 ----------------------- [1723c]
----------------------- [1723c] 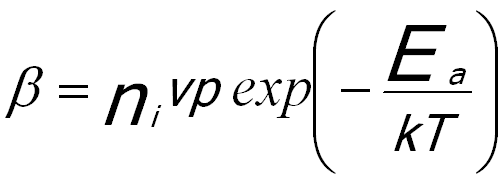 ----------------------- [1723d]
----------------------- [1723d]