|
This book (Practical Electron Microscopy and Database) is a reference for TEM and SEM students, operators, engineers, technicians, managers, and researchers.
|
=================================================================================
Figure 2868a presents the plasmon energy Ep and the energies of relevant core-loss transitions. For light elements (Z<20), the core-loss energies are relatively large (hundreds of eV) and thus do not significantly affect the total EELS intensity. In this case, the scattering is dominated by plasmons and the characteristic EEL energy EC is close to Ep. For heavy elements (Z>20), the contribution of the low-energy core-loss transitions becomes significant thus the difference between EC and Ep is increased. The largest difference is located at Z~29, 48, and 79 for the elements with completed outer d shells, but those atoms do not show low-energy core-loss edges.
The characteristic EEL energies EC is given by, [1]
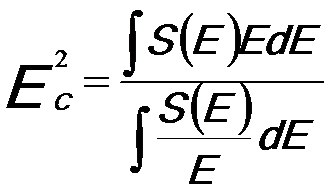 ------------------------ [2868] ------------------------ [2868]
where,
S(E) -- The single-scattering EEL spectrum.
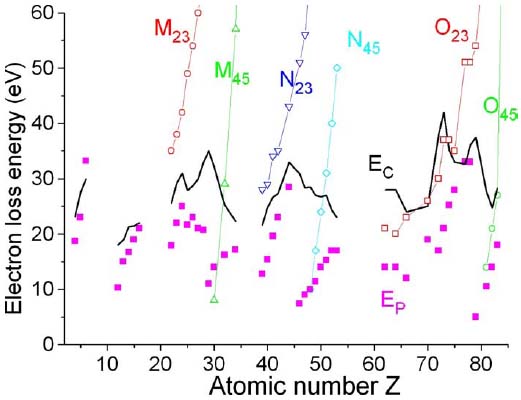
Figure 2868a. Energy of the core-loss (open symbols), energy of plasmon (Ep) transitions (solid squares), and a characteristic EEL energy EC (black solid line).
[1]
Figure 2868b shows the plasmon and low-energy core-loss peaks of yttrium (Y) and palladium (Pd). For the case of Y, the plasmon and core-loss peak can well be separated by spectra fitting with a polynomial function, while for the case of Pd with completed d shells its plasmon peak (~8 eV) is accompanied by a number of other features and their separation can hardly be achieved unambiguously. Those features of Pd probably correspond to different excitations of the composite outer shell consisting of different 4d+5s+5p electronic configurations [1]. Therefore, this interpretation suggests that the number of valence electrons per atom involved in plasmon scattering is larger than number of electrons taking part in chemical reactions.
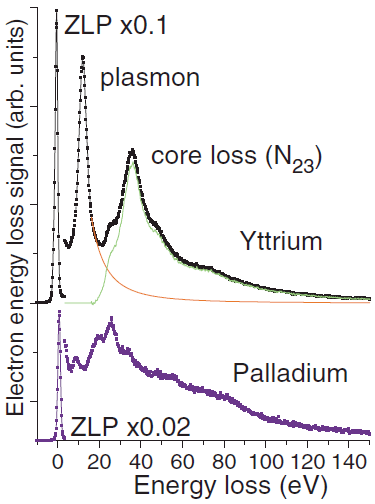
Figure 2868b. Two representative examples (for Y and Pd) of separating the plasmon and low-energy core-loss contributions in the EEL spectra.
[1]
[1] Konstantin Iakoubovskii, Kazutaka Mitsuishi,Yoshiko Nakayama, and Kazuo Furuya, Mean free path of inelastic electron scattering in elemental solids and oxides using transmission electron microscopy: Atomic number dependent oscillatory behavior, Physical Review B 77, 104102 (2008).
|
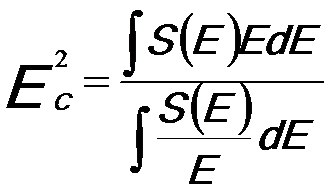 ------------------------ [2868]
------------------------ [2868] 
