=================================================================================
The energy transfer from a charged particle to matter in each individual atomic interaction is normally small and thus, the particle undergoes a large number of interactions before its kinetic energy is completely lost.
In the case of an incident electron beam irradiates a specimen in TEM, the beam can be described by a plane wave. Figure 4430 shows the schematic illustration of momentum transfer and scattering angle of the incident electron beam in an atomic ionization process. Before the interaction of the incident beam with the specimen, the beam has a wave vector k0 (=2π/λ0) and an energy E0. This beam is scattered through an angle θ and loses energy E due to the ionization process. After the interaction it has a wave vector k and a smaller energy E' (=E0 - E). The momentum transferred to the specimen is q (=k0 - k). Their relationship is given by,
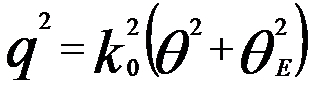 ------------------------------- [4430a] ------------------------------- [4430a]
where,
θE -- The characteristic scattering angle (or called most probable scattering angle) for the given energy loss E.
θE is given by
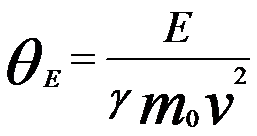 (For general expression) ------------------- [4430b] (For general expression) ------------------- [4430b]
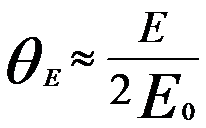 (For an energy of incident electrons is less than 100 keV) ------------------- [4430c] (For an energy of incident electrons is less than 100 keV) ------------------- [4430c]
Where,
γ -- The relativistic factor (=(1 - v2/c2)-1/2)
m0 -- The rest mass of the electron
v -- The velocity of the incident electron
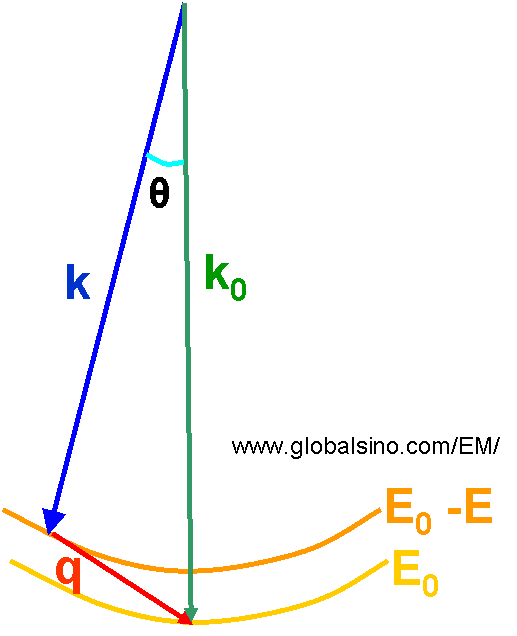
Figure 4430. schematic illustration of momentum transfer and scattering angle of the incident
electron beam in an atomic ionization process.
In EELS measurements, the characteristics originated from the transition of core electrons to unoccupied states in the conduction band require the energy transfer between the incident and core electrons to be greater than its binding energy. The core loss edges are normally represented by a sudden increase in intensity followed by a decrease in intensity with the increase of energy loss. This sudden rise in intensity in EELS profile represents the ionization threshold, the energy of which is approximately equal to the inner-shell binding energy and thus is characteristic of the element.
|
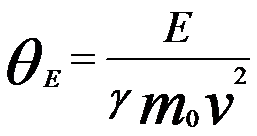 (For general expression) ------------------- [4430b]
(For general expression) ------------------- [4430b] 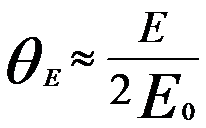 (For an energy of incident electrons is less than 100 keV) ------------------- [4430c]
(For an energy of incident electrons is less than 100 keV) ------------------- [4430c] 