=================================================================================
Table 2026a. Properties of copper (Cu).
Resistivity (10-6 Ω•cm) |
1.67 |
|
Corrosion in air |
Poor |
Melting point (°C) |
1085 |
|
Specific heat capacity(Jkg-1K-1)
|
380 |
Thermal conductivity (Wcm-1) |
3.98 |
|
|
Poor |
Thermal stress per degree for
films on silicon (107 dyn cm-2 °C-1) |
2.5 |
|
Youngs modulus
(10-11 dyn cm-2)
|
12.98 |
Work Function (eV) |
4.65 |
|
|
1.90 |
Coefficient of thermal expansion (CTE)
(10-6 °C-1) |
17 |
|
Density (g-cm-3) |
8.96 |
Heat conductivity (kh,Wcm-1K-1) |
3.94 |
|
Vaporization temperature (Tv, °C) |
2595 |
Melt heat (Qm, J-g-1) |
212 |
|
Vaporization heat (Qv, J-g-1) |
4770 |
Absorption (1-R) |
0.06 |
|
Surface free energy (ergs/cm2) |
850 at 1000 °C |
Electronic configuration |
[Ar] 3d10 4s1 = 1s2 2s2 2p6 3s2 3p6 3d10 4s1 |
|
Crystal system |
FCC |
Kα X-ray for XRD measurements
|
Wavelength (λ) = 0.15418 nm |
|
Oxidation States |
Most common: +1 and +2; some cases: +3 and +4 |
Table 2026b. Some surface energies (J/m2) of low-index surfaces of Cu crystals.
Solid
|
(100)
|
(110)
|
(111) |
(311) |
(511) |
Cu |
1.62 |
1.66 |
1.55 |
1.82 |
1.68 |
Figure 2026a shows the atomic structure of Cu.
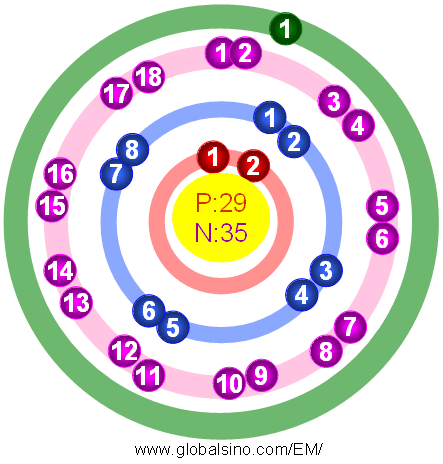
Figure 2026a. Atomic structure of Cu.
Figure 2026b shows the solid solubility of some impurities (including Cu) in silicon.
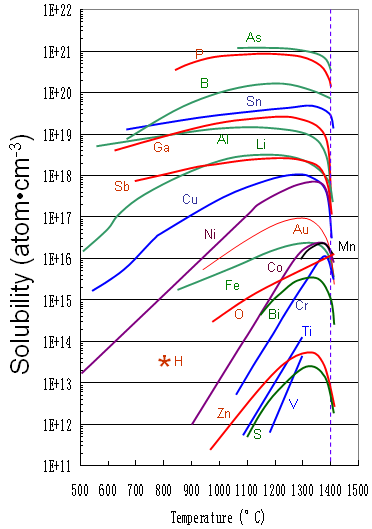
Figure 2026b. Solid solubility of impurities in silicon.
Figure 2026c shows the solubility for impurities (including Cu) in SiC.
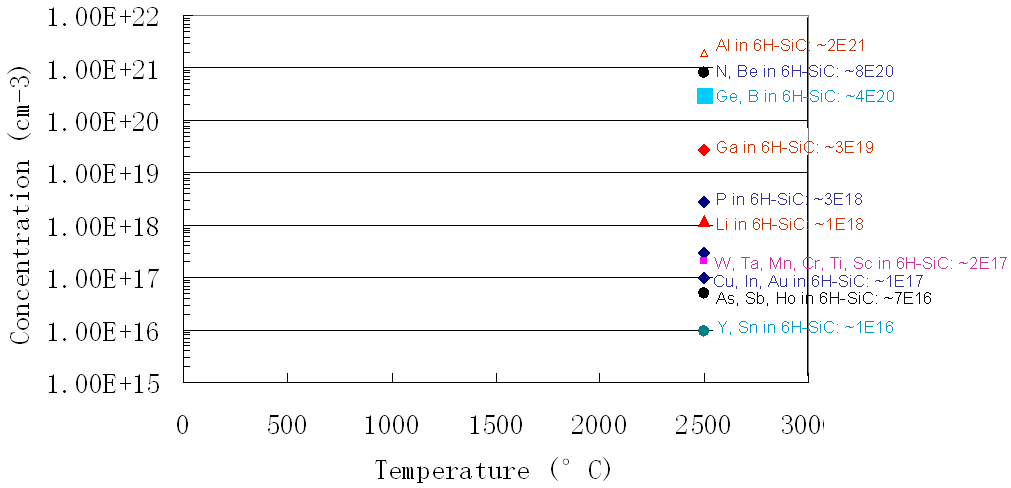
Figure 2026c. Solubility for impurities in SiC.
Table 2026c. Some copper-based alloys.
Alloys |
Page on EM online book |
| Al87Ni7Cu3Ce3 |
page1433 |
|
