=================================================================================
Table 2372a. Properties of Ni metal.
Work Function (eV) |
5.15 |
Electronegativity |
1.91 |
Density (g-cm-3) |
8.9 |
Specific heat (cp, Jkg-1K-1)
|
440 |
Heat conductivity (kh,Wcm-1K-1)
|
0.92 |
Melting temperature (Tm, °C)
|
1453 |
Vaporization temperature (Tv, °C)
|
2840 |
|
309 |
Vaporization heat (Qv, J-g-1)
|
6450 |
Absorption (1-R) |
0.28 for 1 µm light |
Figure 2372a shows the solid solubility of some impurities (including Ni) in silicon.
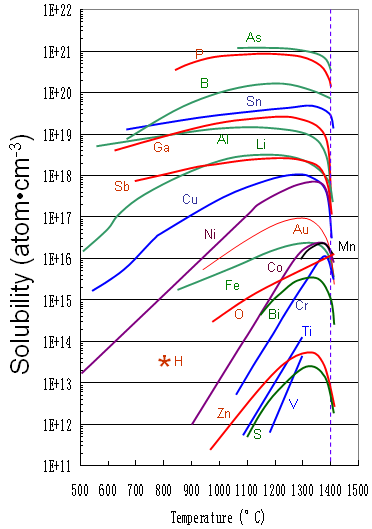
Figure 2372a. Solid solubility of impurities in silicon.
Figure 2372b shows the pair distribution function
(PDF) of crystalline nickel (Ni) simulated with molecular dynamics (MD) model for two different temperatures. In general, the main peaks at higher temperatures shifts to higher r.
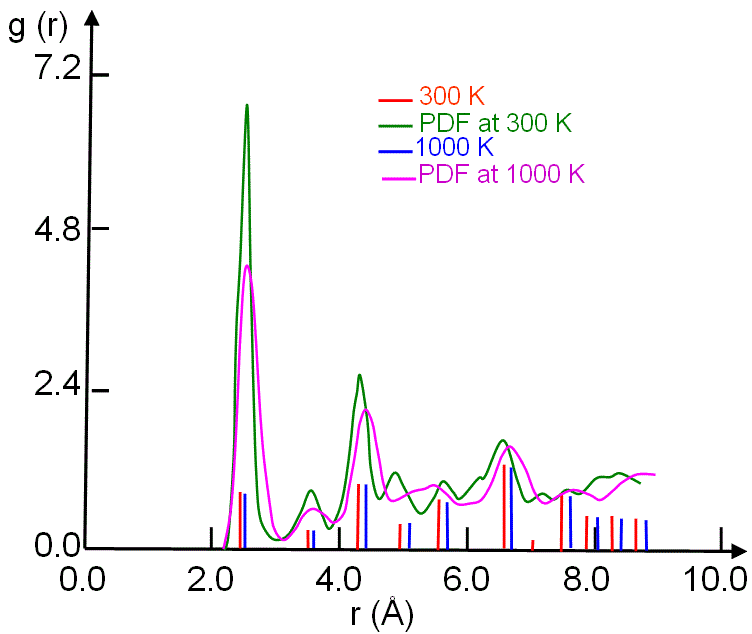
Figure 2372b. PDF of crystalline Ni simulated with molecular dynamics model. Adapted from [1]
Table 2372b. Some nickel-based alloys.
[1] J. Lu and J. A. Szpunar, Phil. Mag. A 75, 1057-1066 (1997).
|