=================================================================================
In EMs, the interactions of the accelerated incident electrons with the specimen result in excitations of electrons into unoccupied energy levels in the conduction band as well as collective excitations of valence electrons. Note that the ionization edges occur at higher energy losses than ~50 eV due to excitation of core electrons into the conduction band.
Figure 3438a shows the schematic illustration of an energy-loss spectrum and the formation of the main energy-loss peaks related to the energy levels of electrons surrounding atom A and atom B in materials.
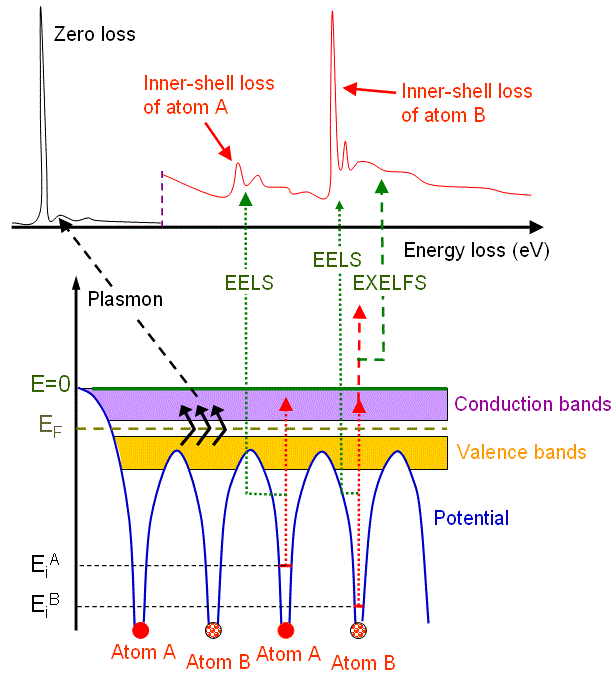
Figure 3438a. Schematic illustration of an energy-loss spectrum and the formation of three main energy-loss peaks.
Fine structures in an EEL spectrum fundamentally originate from the transition from a core level to unoccupied states under the dipole selection rule. The excitation of atomic inner shells enables us to study the unoccupied conduction states in a solid. These core-level processes are mostly sensitive to the final states since the initial states have narrow energy widths. Besides the ionization edges there is a fine structure superposed on the edge and extending up to ~ 50 eV from the EELS edge onset, reflecting the density of unoccupied states in the conduction band and known as the energy loss near edge structure (ELNES).
The characteristics originated from the transition of core electrons to unoccupied states in the conduction band require the energy transfer between the incident and core electrons to be greater than its binding energy. The core loss edges are normally represented by a sudden increase in intensity followed by a decrease in intensity with the increase of energy loss. This sudden rise in intensity in EELS profile represents the ionization threshold, the energy of which is approximately equal to the inner-shell binding energy and thus is characteristic of the element.
Figure 3438b shows the measured CeF3 F-K ELNES. [1] The main structure width, including the large peak A and its shoulder B, was evaluated as the distance between the positions of the broken lines corresponding to the minimum and maximum in the second differentiated spectra.
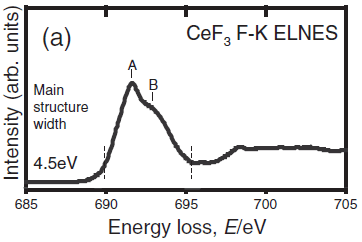
Figure 3438b. The measured CeF3 F-K ELNES. Adapted from [1]
The core-loss edges in EELS are a projection of the atom resolved, partial density of states (PDOS) of the conduction band. Figure 3438b shows the unoccupied partial density of states (PDOS) of CeF3 and the bond overlap population diagram (BOPD) between Ce and its neighboring F for the non-core-holed, ground state electronic structures. The main peak structures of the ELNES in Figure 3438b correspond to the Ce5d derived states, which range from 5 to 9 eV above the Fermi level (the Fermi level in Figure 3438c is set to zero). Since the intensities of the BOPD are negative, the corresponding interactions between the Ce and anion F atoms are all antibonding. Larger BOPD intensities correspond to larger spatial overlaps between the two molecular orbitals, promoting higher antibonding energy levels. [2] The spatial overlaps between the Ce5d and the F orbitals show a variation in the Ce5d-derived states, resulting in the energy width of Ce5d PDOS and the main peak structures in the ELNES in Figure 3438b.
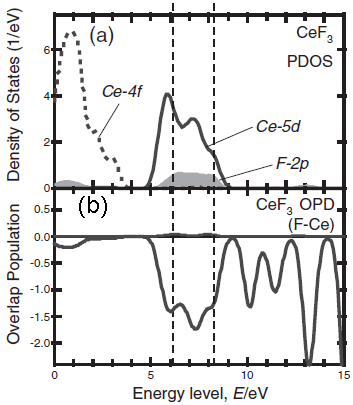
Figure 3438c. The unoccupied PDOS of CeF3 and the BOPD between Ce and its neighboring F for the non-core-holed, ground state electronic structures. Adapted from [1]
[1] Ikuo Nishida, Kazuyoshi Tatsumi and Shunsuke Muto, Local Electronic and Atomic Structure of Ce3+-Containing Fluoride/Oxide Determined by TEM-EELS and First-Principles Calculations, Materials Transactions, Vol. 50, No. 5 (2009) 952 - 958.
[2] D. G. Pettifor: Bonding and Structures of Molecules and Solids,
(Oxford University Press, 1995).
|

