|
|
Rhenium Trioxide (ReO3)
- Practical Electron Microscopy and Database -
- An Online Book -
|
|
http://www.globalsino.com/EM/
|
|
This book (Practical Electron Microscopy and Database) is a reference for TEM and SEM students, operators, engineers, technicians, managers, and researchers.
|
=================================================================================
Rhenium trioxide (ReO3) is a highly insoluble thermally stable material suitable for glass, optic and ceramic applications. This oxide is a chemical compound which can be formed when rhenium oxidizes in air. It is not electrically conductive.
ReO3 forms crystals with a primitive cubic unit cell, with a lattice parameter of ~0.39 nm. The structure of ReO3 is similar to the perovskite structure (ABO3), without the large A cation at the centre of the unit cell. Each rhenium atom is surrounded by an octahedron of six oxygen centers. Therefore, as shown in Figure 3353, the ReO3 structure consists of a cubic array of corner-sharing ReO6 octahedra with square-shaped tunnels running in the direction of the main crystallographic axes. The coordination number of O is 2 because each oxygen atom has 2 neighbouring Re atoms.
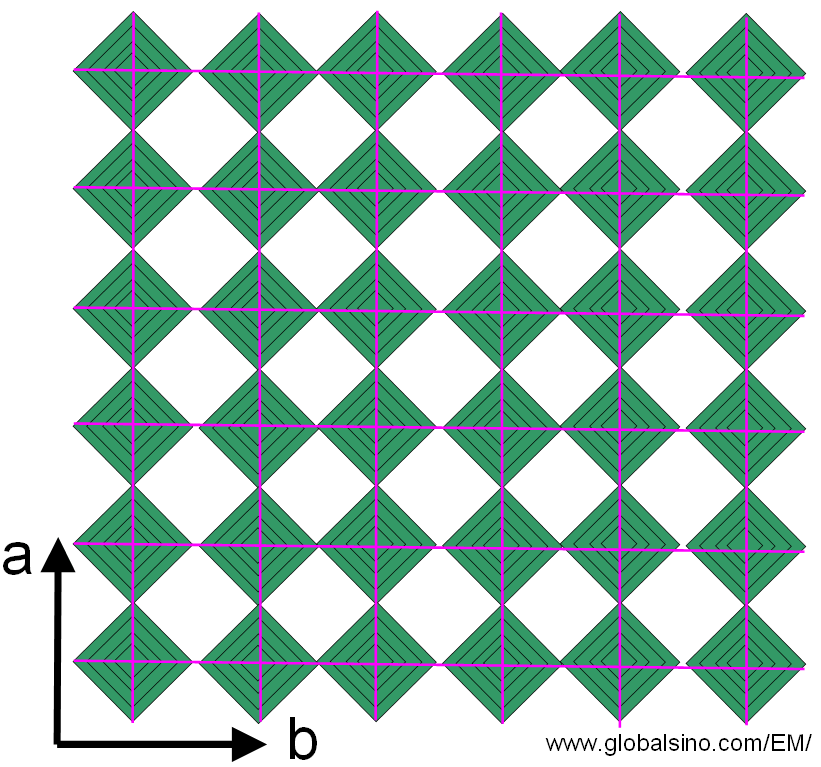
Figure 3353. Model projected onto the ab-plane of ideal ReO3 structure.
Two other structure types are closely related to the ReO3 type, i.e. the block structures and the tetragonal tungsten bronzes (TTB).
|
=================================================================================
The book author (Dr Y. Liao) welcomes your comments, suggestions, and corrections, please click here for submission. You can click How to Cite This Book to cite this book. If you let Dr Y. Liao know once you have cited this book, the brief information of your publication will appear on the “Times Cited” page.
|
|
