=================================================================================
Table 3427a. Main edges of vanadium (V) used in EELS analysis.
Edge(s) |
Edge onsets (eV) |
| L3,2 |
513 |
Most EELS studies of vanadium (V) have been performed at its L3/L2 edge.
Pearson et al. [1] experimentally and theoretically (based on one-electron Hartree-Slater calculations) found that the intensities of L2,3 white lines for most of the 3d and 4d transition metals decreased nearly linearly with increasing atomic number, reflecting the filling of the d states. Figure 3427a shows the deconvoluted and background-subtracted L2,3 energy-loss spectra for the 3d transition metals. The edge energies are not shown in order to present all the spectra in the same figure, while the intensities of the white lines are scaled simultaneously for all elements.
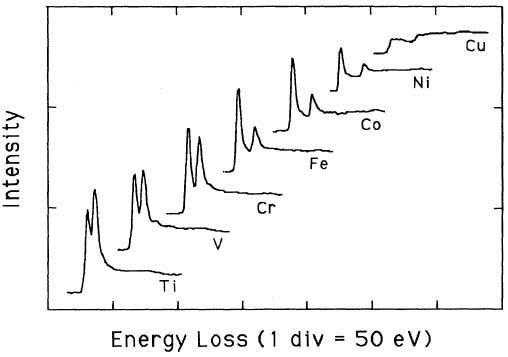
Figure 3427a. The deconvoluted and background-subtracted L2,3 energy-loss spectra for the 3d transition metals. [1]
Figure 3427b shows EEL spectra of TiO2, V, Cr, Fe2O3, CoO, NiO, and Cu with L2,3 energy-losses for the 3d transition metals. The chemical shift of O element in the different oxides (TiO2, Fe2O3, CoO, and NiO) is also indicated in the figure. The main edges of Ti, V, Cr, Fe, Co, Ni and Cu, and their edge onsets are listed in Table 3427b.
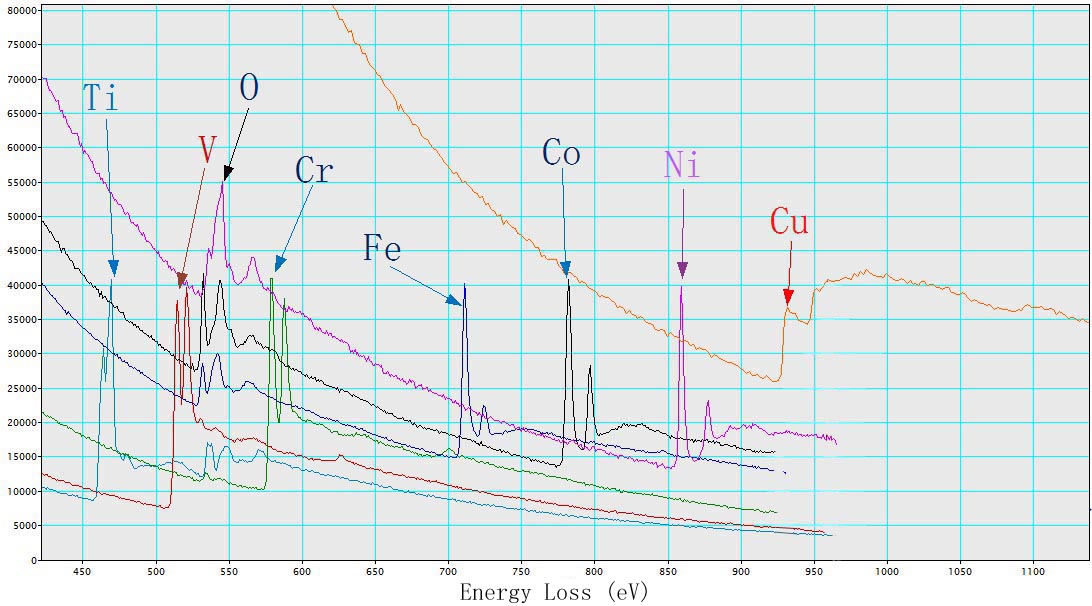
| Figure 3427b. EEL spectra of TiO2, V, Cr, Fe2O3, CoO, NiO, and Cu. |
Table 3427b. Main edges of Ti, V, Cr, Fe, Co, Ni, and Cu, and their edge onsets.
| Atomic number |
Element |
Edge(s) |
Edge onsets (eV) |
| 22 |
Ti |
L2,3 |
455 |
| 23 |
V |
L2,3 |
513 |
| 24 |
Cr |
L2,3 |
575 |
| 26 |
Fe |
L2,3 |
710 |
| 27 |
Co |
L2,3 |
779 |
| 28 |
Ni |
L2,3 |
855 |
| 29 |
Cu |
L2,3 |
935 |
In order to quantify the correlation between the intensities of the white lines and the d holes (or occupancy), the intensities of the white lines are normally obtained by subtracting the background from the signal. Figure 3427c illustrates the method for isolating and normalizing the white lines for 3d transition V (vanadium) metal. The background intensity is modeled by double step functions in the threshold regions. A straight line over a range of ~ 50 eV is fit to the background intensity immediately following white line L2. A linear correlation of the normalized white line intensity, I3d, with 3d occupancy, n3d (see Tables in page4697), is obtained with a fit given by,[1]
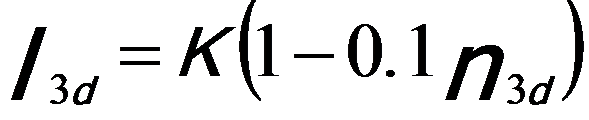 -------------------------- [3427a] -------------------------- [3427a]
where, the constant, K, is arbitrary and depends on the specific normalization scheme.
By fitting the constant with the experimental data, the normalized white-line intensities for steps at peaks (Equation 3427b) and at onsets (Equation 3427c) can be given by, [1]
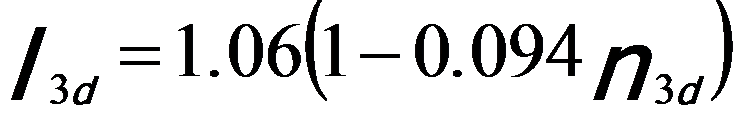 -------------------------- [3427b] -------------------------- [3427b]
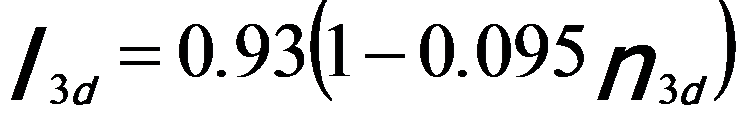 -------------------------- [3427c] -------------------------- [3427c]
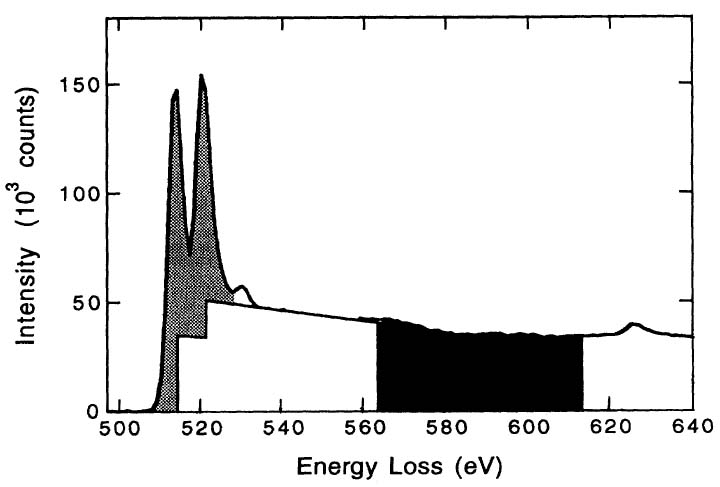
Figure 3427c. Schematic illustration of L2,3 edges of V transition metal for
background subtraction and intensity normalization. [1]
In the determination of the stoichiometry of the vanadate oxides, the researchers have mainly focused on the
high energy loss regions, including the vanadium L2,3 peaks and oxygen K peak, for instance shown in Figure 3427d. The vanadium L2 and L3 peaks denote electronic transitions from vanadium 2p1/2 and 2p3/2 states to the orbitals with 3d state [3]. The oxygen k-edge (between 525 and 550 eV) presents the transition from the oxygen 1s state to the orbitals with oxygen 2p state. Note that care is needed when we are doing mapping analysis or quantification, e.g. using three-window method, since the V edges and O edge are very close in energy.
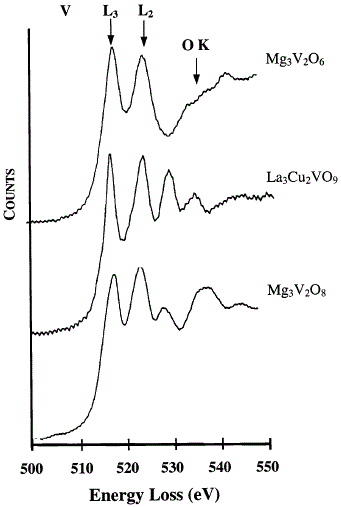
Figure 3427d. EEL spectra: (a) Mg3V2O6, (b) La3Cu2VO9 and (c) Mg3V2O8. [2]
Table 3427c presents the ratios of the EELS L3/L2 edges of La3Cu2VO9, Mg3V2O6 and Mg3V2O8 where vanadium exhibits the +5, +3 and +5 oxidation states, respectively. The L3/L2 ratio obtained for La3Cu2VO9 is very close to that of Mg3V2O8 because they have the same oxidation states.
Table 3427c. Measured ratios between background-subtracted,
integrated intensities of V L3 and V L2 peaks.
| Oxide |
V L3/L2 ratio |
Oxidation state |
Reference |
| Mg3V2O6 |
0.90(8) |
+3 |
[4] |
| Mg3V2O8 |
0.72(4) |
+5 |
[4] |
| La3Cu2VO9 |
0.69(6) |
+5 |
[2] |
[1] D. H. Pearson, C. C. Ahn, and B.Fultz, White lines and d-electron occupancies for the 3d and 4d transition metals, Physical Review B, 47(14), (1993) 8471-8478.
[2] Sylvie Malo, Douglas A. Vander Griend, Kenneth R. Poeppelmeier, Yanguo Wang, Vinayak P. Dravid, Crystal symmetry of La3Cu2VO9 and La4Cu3MoO12 derived from the YAlO3 hexagonal structure by transmission electron microscopy, Solid State Sciences 3 (2001) 17–23.
[3] C.C. Ahn, O.L. Krivanek, EELS Atlas (Center for Solid
State Science, Arizona State university, Tempe, AZ) (1983).
[4] X. Wang, H. Zhang, W. Sinkler, K.R. Poeppelmeier, L.D.
Marks, J. Alloys Comp. 270 (1998) 88.
|