EELS Measurement of Chromium (Cr) - Practical Electron Microscopy and Database - - An Online Book - |
||||||||||||||||||||||||||||||||||||||||||
| Microanalysis | EM Book http://www.globalsino.com/EM/ | ||||||||||||||||||||||||||||||||||||||||||
Table 3428a. Main edges of Cr used in EELS analysis.
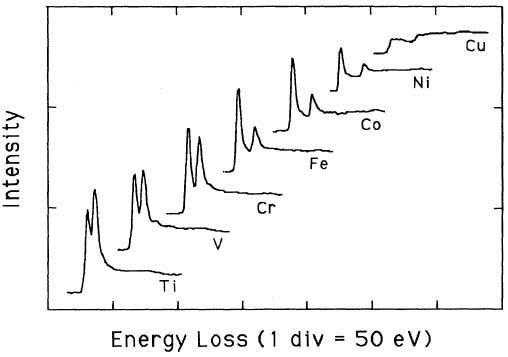
Pearson et al. [1] experimentally and theoretically (based on one-electron Hartree-Slater calculations) found that the intensities of L2,3 white lines for most of the 3d and 4d transition metals decreased nearly linearly with increasing atomic number, reflecting the filling of the d states. Figure 3428a shows the deconvoluted and background-subtracted L2,3 energy-loss spectra for the 3d transition metals. The edge energies are not shown in order to present all the spectra in the same figure, while the intensities of the white lines are scaled simultaneously for all elements.
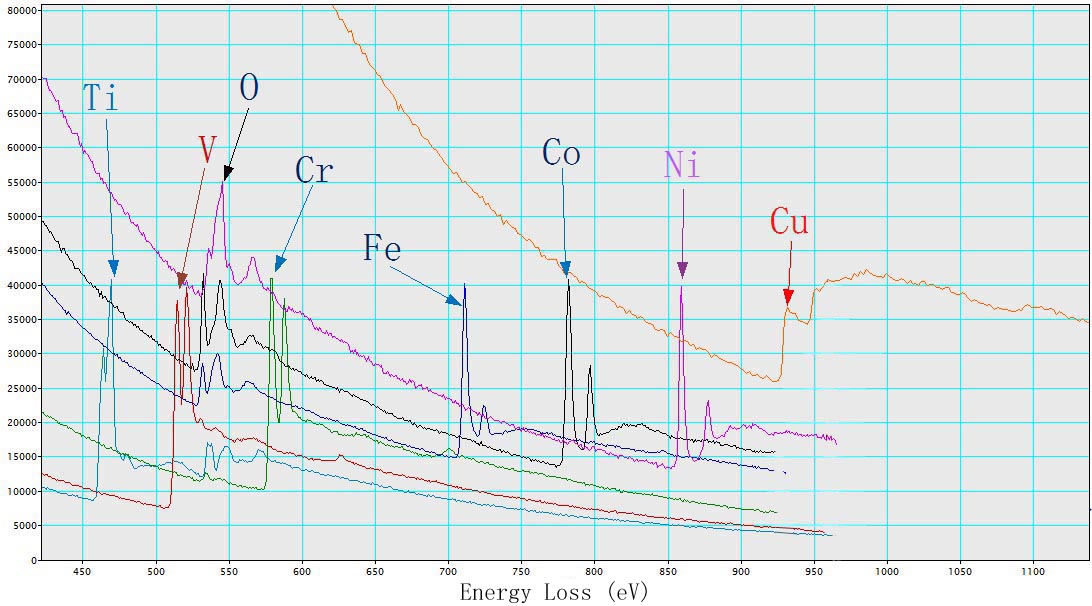
Figure 3428a. The deconvoluted and background-subtracted L2,3 energy-loss spectra for the 3d transition metals. [1] Figure 3428b shows EEL spectra of TiO2, V, Cr, Fe2O3, CoO, NiO, and Cu with L2,3 energy-losses for the 3d transition metals. The chemical shift of O element in the different oxides (TiO2, Fe2O3, CoO, and NiO) is also indicated in the figure. The main edges of Ti, V, Cr, Fe, Co, Ni and Cu, and their edge onsets are listed in Table 3428b.
Table 3428b. Main edges of Ti, V, Cr, Fe, Co, Ni, and Cu, and their edge onsets.
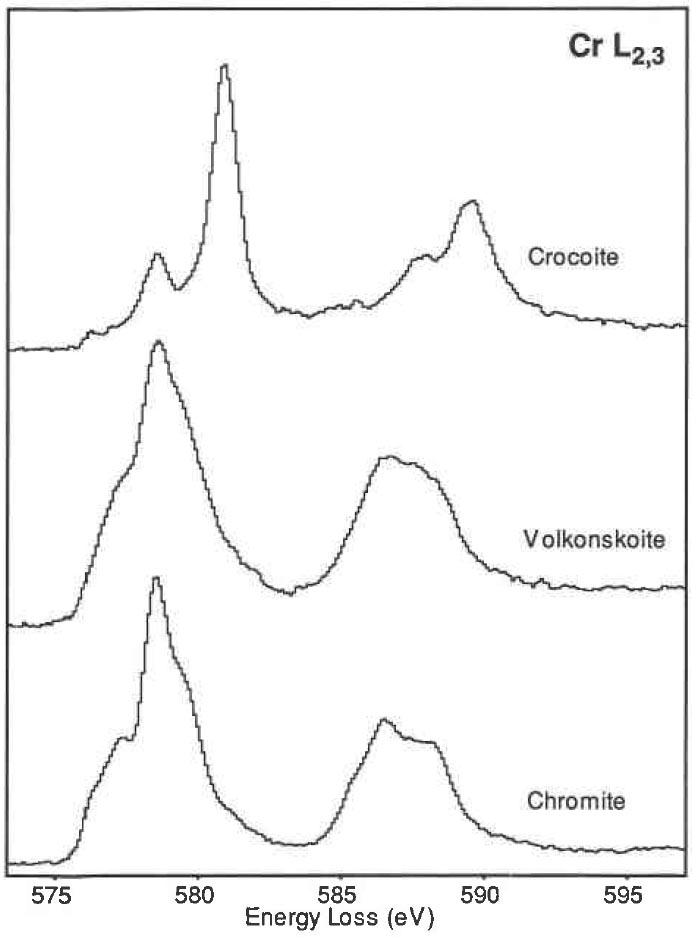
The L2,3 ELNES of Cr (Cr3+ and Cr6+) shows multiplet structures of valences representing valence fingerprints. Figure 3428c shows Cr L2,3 edges obtained from chromite (Cr3+), volkonskoite (Cr3+), and crocoite (Cr6+).
Figure 3428c. Cr L2,3 edges obtained from chromite (Cr3+), volkonskoite (Cr3+), and crocoite (Cr6+). For elemental mapping of oxygen (O) and chromium (Cr) with their closely spaced ionization edges, multiple linear least-squares (MLLS) fitting [2] and four window method [3] had been proposed to map Cr with the L2,3-edge in oxidized specimens.
[1] D. H. Pearson, C. C. Ahn, and B.Fultz, White lines and d-electron occupancies for the 3d and 4d transition metals, Physical Review B, 47(14), (1993) 8471-8478.
|
|
|||||||||||||||||||||||||||||||||||||||||