EELS Measurement of Aluminum (Al) - Practical Electron Microscopy and Database - - An Online Book - |
||||||||||||
| Microanalysis | EM Book http://www.globalsino.com/EM/ | ||||||||||||
Table 3446. Main edges of Al used in EELS analysis.
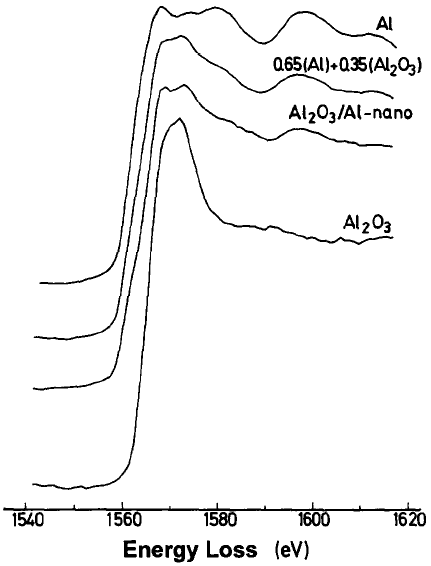
Some inelastic mean free paths (λ) of incident electrons at different kinetic energies in metallic Al and Al-based components are given in Table 4623. On the other hand, the λ in Al2O3 at kinetic energies 500 eV and 2 keV are 12.9 Å and 36.7 Å, respectively. [4] Figure 3446a shows the Al near K-edge structure of the EELS spectra (ELNES) obtained in TEM from the samples: i) Al foil; ii) 35% of Al in the form of A12O3 and 65% of metallic Al; iii) A12O3/Al-nano sample (mean particle size of ~ 23 nm); and iv) An alumina (A12O3).
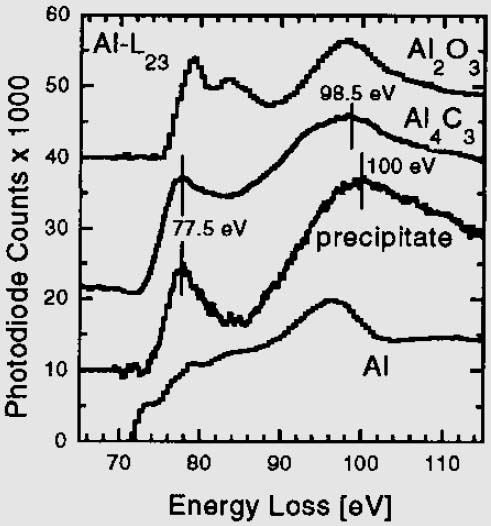
Figure 3446a. Al near K-edge structure of the EELS spectra (ELNES) obtained in TEM from the samples with different Al and A12O3 materials. [1] Figure 3446b shows the Al L2,3 ELNES profile of A12O3, Al4C3, precipitate in C/Mg-Al composites, and metallic Al. In both cases of Al4C3 and precipitate in C/Mg-Al composites, the edge-onset energy is at ~ 73 eV, followed by a peak at ~ 77.5 eV.
Figure 3446b. The Al L2,3 ELNES profiles of different Al compounds. [3]
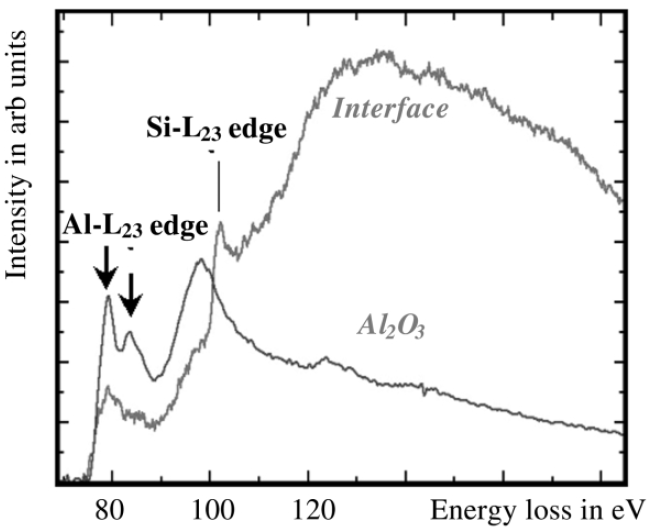
It is necessary to note that the Al L2,3 edge and Si L2,3 edge in EEL spectra are very close as shown in Figure 3446c.
Figure 3446d shows the structure of Al L2,3 edges obtained from an interface between Si (silicon) and Al (aluminum) layers, and from an Al2O3 (aluminum oxide) materials. The Al L2,3 ionisation edge are the same in both cases. The two maxima at approximately 79 and 84 eV are typical peaks from Al2O3 materials.
Figure 3446d. Al L2,3 ELNES (energy loss near-edge structure) profiles taken from a Si/Al interface and from an Al2O3 TEM film.
[2]
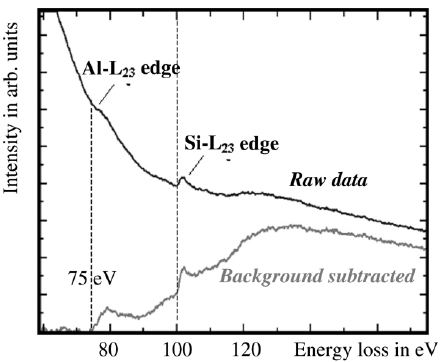
The EEL spectra in Figure 3446e show the Al L2,3 edge as well as the Si L2,3 edge after background subtraction, taken from AlxSiyOz matrix. The threshold value of the Al ionization edge here is about 75 eV indicating that aluminum is bonded to oxygen because the onset energy of the edge of pure aluminum is about 73 eV. This energy difference is called chemical shift.
Figure 3446e. EELS profile of Al and Si taken from AlxSiyOz matrix by a electron probe of 2 nm in diameter. [2]
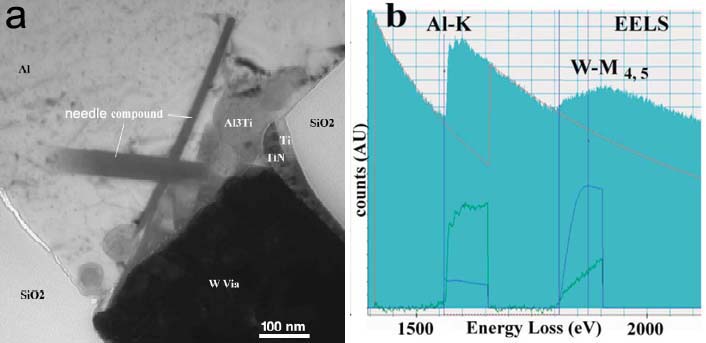
Figure 3446f (a) shows a typical bright field TEM image of an Al/Ti/W/TiN via after thermal stress at 450 °C for 4 hours. An Al3Ti layer is formed between Al and W via. Figure 3446f (b) shows an EELS profile acquired over 20 s in the needle inclusion. The chemical formula of the needles is between Al10W and Al12W.
Figure 3446f. (a) A typical bright field TEM image of an Al/Ti /W/TiN via after thermal
[1] J. C. Sánchez-López, A. Caballero and A. Fernández, Characterisation of Passivated Aluminium Nanopowders: An XPS and TEM/EELS Study, Journal of the European Ceramic Society 18 (1998) 1195 - 1200. |
|
|||||||||||