=================================================================================
Two types of X-rays are induced by the electron beam: characteristic X-rays and bremsstrahlung X-rays as shown in Figure 4688a. A continuum of bremsstrahlung X-rays is produced in the low energy regimes if an electron is decelerated by the nucleus. However, the bremsstrahlung can actually be any value from 0 up to the incident electron energy. The bremsstrahlung X-rays are normally broad and are featureless background to characteristic X-rays. Bremsstrahlung X-rays are useful and convenient to the biologist, but usually annoys most materials scientists.
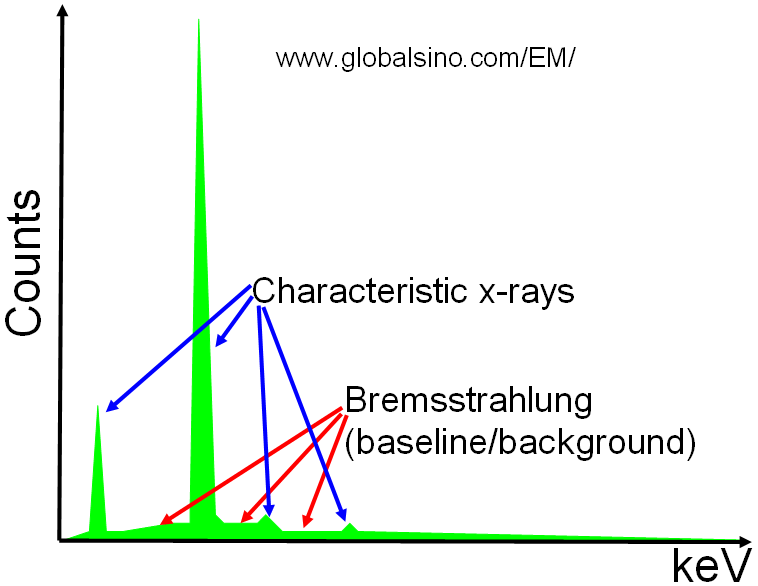
Figure 4688a. Schematic showing X-rays of a sample element.
The charged particle experiences Coulomb interactions with the nuclei and the orbital electrons of the atoms in a matter when it travels through the matter. As shown in Figure 4688b, these interactions can be divided into three categories depending on the impact parameter b compared with the atomic radius a: a) Interaction with the external nuclear field (bremsstrahlung production) for b << a; b) Interaction with the orbital electrons for b ≈ a (hard collision); c) Interaction with the orbital electron for b >> a (soft collision).
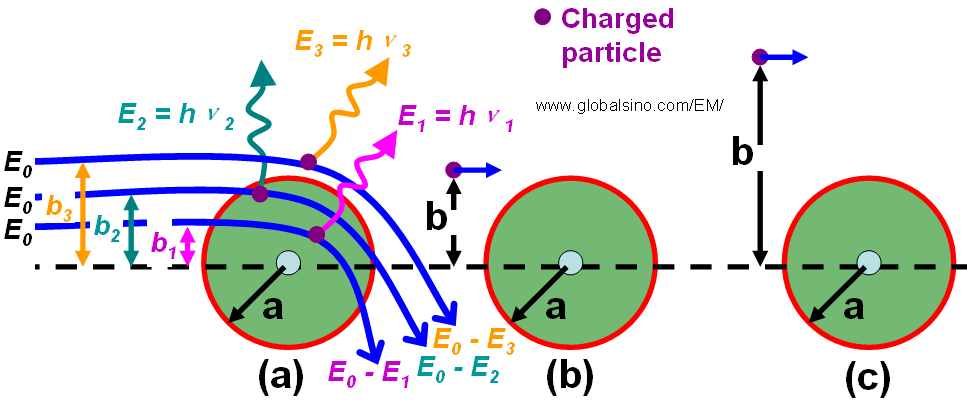
Figure 4688b. Radiative (a), hard (b) and soft (c) collisions. b is the impact parameter and a is the atomic radius.
Table 4688 shows that electrons interact with 1 electron, many electrons, 1 nucleus, and many nuclei in solids.
Table 4688. Effects of interactions of electrons in solids.
| |
Interaction with electron(s) |
Interaction with nucleus/nuclei |
| |
1 electron |
Many electrons |
1 nucleus |
Many nuclei |
| Scattering type |
Inelastic |
Inelastic |
Quasi-elastic |
Elastic |
Inelastic |
| Scattering effect |
Electron Compton effect; electron excitation (from 50 eV to a few keV: EDS and EELS) |
Plasmon excitation (< 50 eV, ~100 nm TEM specimen); Cerenkov effect |
Rutherford scattering; phonon scattering (< 1 eV, heat) |
Bragg scattering |
Bremsstrahlung |
The intensity of the bremsstrahlung x-rays (Ib) at an energy (Ev) is quantified by Kramers' law,
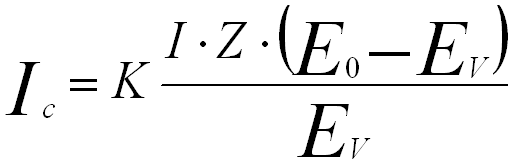 ---------------------- [4688] ---------------------- [4688]
where,
Z -- The average atomic number of the specimen,
E0 -- The incident beam energy,
I -- The electron beam current,
Ev -- The continuum photon energy.
Based on Equation 4688, samples with higher atomic number generate proportionately
more bremsstrahlung background. The x-ray continuum intensity decreases with increase of the photon energy, yielding zero at the energy of the incident electron beam. At low X-ray photon energies the intensity increases rapidly due to the greater probability for slight deviations in the trajectory caused by the Coulombic field in the atoms.
Note that, when EM samples are nonconductive, negative charging of the sample surface will cause a shift of the maximum energy of the Bremsstrahlung
background to lower energies.
The main inelastic scattering mechanisms are:
i) Phonon excitation (heat).
ii) Plasmon excitation (valence electrons).
iii) Single electron excitation (inner and outer shell scattering).
iv) Direct radiation losses (Bremsstrahlung radiation due to deceleration of the electron beam in the Coulomb field of an atom).
v) Excitation of conducting electrons leading to secondary electron emissions.
The bremsstrahlung background forms a smooth continuum with count rate per unit energy interval that is independent of detector resolution. Furthermore, EDS has relatively low peak-to-background ratio in electron microscopes due to the high background coming from bremsstrahlung radiation emitted by electrons suffering deceleration on scattering by atoms. This background continuum makes it unable to detect characteristic X-rays from components below the 0.1-0.05% level. Even though trace elements down to ~500 ppm of oxides can be detected by SEM-EDS, their quantification is not accurate. Note that collecting EDS data for longer time only slightly improves the detection limit.
|