=================================================================================
Energy-filtered transmission electron microscopy (EFTEM) is a technique used in transmission electron microscopy, in which only electrons of particular kinetic energies are used to form the image or diffraction pattern (See EFTEM System and Operation Principle). In this case, instead of sweeping the spectrum over the slit in serial EELS, the magnetic field is held constant and a single energy window is selected, and then the energy-filtered image is formed. This technique can be used to aid chemical analysis of the sample in conjunction with complementary techniques such as electron crystallography. EFTEM can also be
used to improve the contrast in images and diffraction patterns by removing inelastically
scattered electrons.
If a very thin sample is illuminated with a beam of high-energy electrons, then a majority of the electrons will pass unhindered through the sample but some will interact with the sample, being scattered elastically or inelastically (phonon scattering, plasmon scattering or inner shell ionisation). Inelastic scattering results in both a loss of energy and a change in momentum, which in the case of inner shell ionisation is characteristic of the element in the sample.
If the electron beam emerging from the sample is passed through a magnetic prism, then the flight path of the electrons will vary depending on their energy. This technique is used to form spectra in Electron energy loss spectroscopy (EELS), but it is also possible to place an adjustable slit to allow only electrons with a certain range of energies through, and reform an image using these electrons on a detector.
The energy slit can be adjusted so as to only allow electrons which have not lost energy to pass through to form the image. This prevents inelastic scattering from contributing to the image, and hence produces an enhanced contrast image.
Adjusting the slit to only allow electrons which have lost a specific amount of energy can be used to obtain elementally sensitive images. As the ionisation signal is often significantly smaller than the background signal, it is normally necessary to obtain more than one image at varying energies to remove the background effect. The simplest method is known as the jump ratio technique, where an image recorded using electrons at the energy of the maximum of the absorption peak caused by a particular inner shell ionisation is divided by an image recorded just before the ionisation energy. It is often necessary to cross-correlate the images to compensate for relative drift of the sample between the two images.
Improved elemental maps can be obtained by taking a series of images, allowing quantitative analysis and improved accuracy of mapping where more than one element is involved. By taking a series of images, it is also possible to extract the EELS profile from particular features.
Energy filtering function is achievable with two types of commercial
energy-filtering TEMs: i) In-column ‘omega filters’ or gamma-filters; ii) Post-column imaging filters in a TEM (e.g. Gatan imaging filter), or with so-called spectrum imaging in a STEM. In these techniques an EELS spectrum is collected at each pixel of a camera. Figure 4993a compares the two commercial fixed-beam energy filters (a typical Zeiss Ω filter and a typical Gatan filter) for energy selected imaging (ESI) in TEM columns. Both can be used in EFTEM imaging.
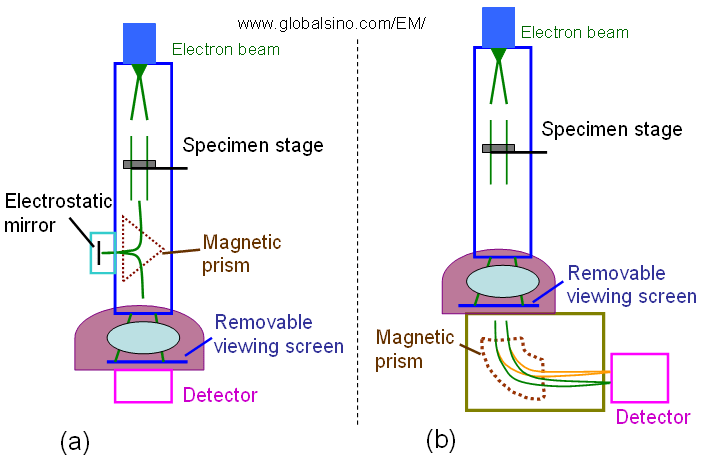
Figure 4993a. Schematic comparison between a typical Zeiss Ω filter (a) and a typical Gatan filter (b) for energy selected imaging (ESI) in TEM columns.
The GIF (Gatan Imaging Filter) is mounted below the column of a TEM. It consists of two main parts:
i) Magnetic prism
In the curved magnetic field, electrons with different energies are dispersed, and consequently an EEL spectrum is obtained at the end of this part. In fact, the magnetic prism is the same as in a parallel electron energy loss spectrometer, and, of course, an EEL spectrum can be measured with the GIF as well.
ii) Optical column
An energy range is selected by a slit located at the dispersion plane. In imaging mode, electrons of the selected energy pass through the lenses of the optical system, and an energy-filtered image occurs at the end of the GIF, where a two-dimensional (2D) slow-scan CCD camera is installed for image recording.
Only electrons scattered at small angles and without loosing energies will contribute to the information in the HRTEM images while all the others (which have lost energy and have been scattered multiple times or at high angles) also contains important information about the sample, they will for instance give a diffuse background in the TEM images and benefit to EFTEM elemental mapping and EELS measurements.
The terms ‘electron spectroscopy imaging’ (ESI) [1] and ‘image spectroscopy’/‘imaging spectrum’ (IS) [2 - 4] have been commonly used for methods acquiring EFTEM series. In contrast, the term ‘spectrum-imaging’ (SI) [5 - 6] is used for acquiring similar three-dimensional data blocks by STEM.
Both EFTEM and spectrum mapping in STEM mode can be used for elemental mapping and ELNES mapping. However, elemental mapping based on energy filter still has some limitations comparing with other methods. For instance, differentiation between CoSi and CoSi2 can be performed using Si L2,3 ELNES mapping but not EELS elemental mapping.
In EFTEM imaging, an important contribution for low-loss imaging is the delocalization of the inelastic scattering process itself, while at much higher energy-losses the resolution-limiting parameter is usually the chromatic aberration. For instance, to lower the effect of the chromatic aberration, the accelerating voltage needs to be 300 kV if one expects a resolution better than 0.3 nm for >100 eV energy-loss [7].
[1] J. Mayer, U. Eigenthaler, J.M. Plitzko, F. Dettenwanger,
Micron 28 (1997) 361.
[2] P.J. Thomas, P.A. Midgley, Ultramicroscopy 88 (2001)
187.
[3] P.J. Thomas, P.A. Midgley, Ultramicroscopy 88 (2001)
179.
[4] J.-L. Lavergne, J.-M. Martin, M. Belin, Microsc. Microanal.
Microstruct. 3 (1992) 517.
[5] C. Jeanguillaume, C. Colliex, Ultramicroscopy 28 (1989)
252.
[6] J.A. Hunt, D.B. Williams, Ultramicroscopy 38 (1991) 47.
[7] R.F. Egerton, Electron Energy-Loss Spectroscopy in the
Electron Microscope, 2nd Edition, Plenum Press, New
York, 1996.
|