|
|
Cleavage Plane of Crystals
- Practical Electron Microscopy and Database -
- An Online Book -
|
|
https://www.globalsino.com/EM/
|
|
This book (Practical Electron Microscopy and Database) is a reference for TEM and SEM students, operators, engineers, technicians, managers, and researchers.
|
=================================================================================
Sapphire (α-Al2O3) as a hexagonal structure, belonging to the space group R3c, can be expressed both as a hexagonal as well as a rhombohedral unit cell. The basic structure consists of hexagonal close-packed planes of oxygen intercalated with aluminum planes. As shown in Figure 2079, the aluminum planes have a similar hexagonal close-packed arrangement but with 1/3 of the sites vacant, resulting in an Al/O ratio of 2/3. Each Al atom is coordinated by 6 oxygen atoms, and each oxygen atom has 4 Al neighbours. The vacancies are aligned on the so-called R-planes, giving sapphire the ability to cleave along these rhombohedral planes. Due to these properties, the unit cell of sapphire is chosen taking into consideration of the position of the A1 vacancies.
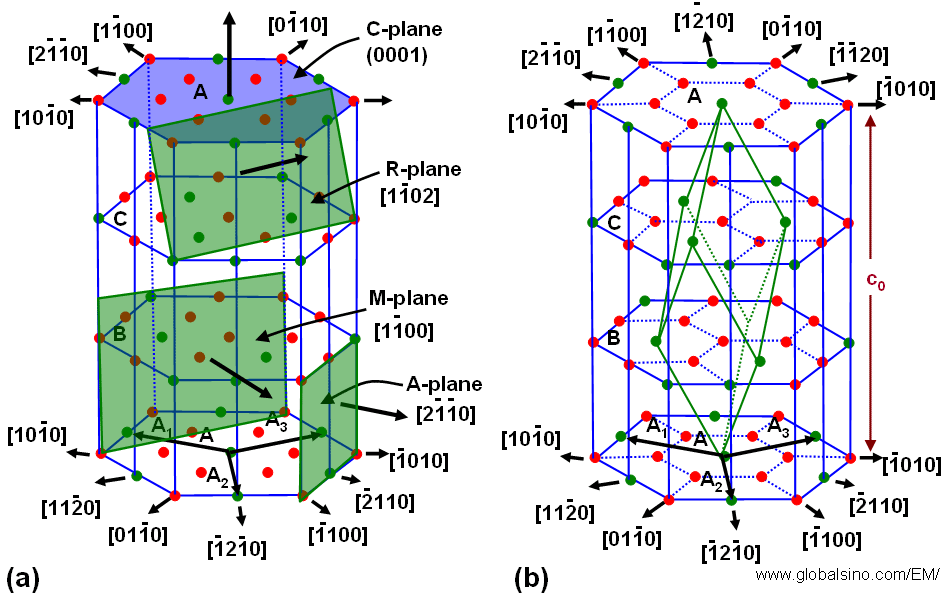
Figure 2079.The crystal lattice of sapphire. (a) A-, M-, R-, and C-planes of the crystal; (b) Schematic illustration of the aluminum planes. Oxygen planes are intercalated with the aluminum planes (not shown).
|
=================================================================================
The book author (Yougui Liao) welcomes your comments, suggestions, and corrections, please click here for submission. You can click How to Cite This Book to cite this book. If you let Yougui Liao know once you have cited this book, the brief information of your publication will appear on the “Times Cited” page.
|
|
