=================================================================================
Rutherford cross-section gives convenient formulas to calculate the elastic scattering angle and total elastic cross section so that it is often used to describe the elastic scattering of electron. However, its accuracy becomes worse in low-energy region or in high atomic-number materials. The Rutherford scattering cross section, or electron-nucleus scattering is given by,
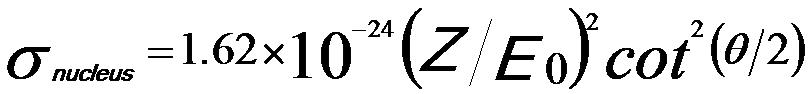 -------------------- [4425a] -------------------- [4425a]
where,
θ -- The scattering angle.
The Rutherford theory predicted the cross section of elastic scattering σ(Z) ∼ Z2. However, it neglected the electronic structure of the scattering atom. Therefore, the Z2 law is only a first approximation. Therefore, we also can have,
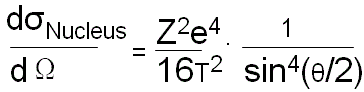 ------------------------------- [4425b] ------------------------------- [4425b]
where,
T -- The kinetic energy of incident electrons.
Furthermore, based on the Rutherford theory above, the total probability of scattering an electron can be estimated (see page2673). The physical mechanism for the Z-dependent behavior of HAADF-STEM contrast is that high-angle scattering is mostly from the electrons scattered by the inner cores of atoms. The partial screening of the atomic nucleus by the inner shell electronic charges is responsible for the value of n to be less than 2 for a pure Rutherford scattering.
|