=================================================================================
The energy of X-ray “particles” (photons) is hν. Here, h is Planck’s constant and ν is the frequency. The X-ray energy in eV is given by,
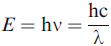 ----------------------- [4484a] ----------------------- [4484a]
The X-ray wavelength, λ, is given by,
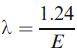 ------------------------------ [4484b] ------------------------------ [4484b]
where λ is in nm and E in keV. As we can see, this equation is similar to the equation giving the uncorrected electron energy in vacuum.
It is very common that Cu X-ray lines are applied to X-ray diffraction (XRD) analysis. The X-ray wavelengths (λ) of Cu Kα1 and Cu Kα2 are 1.540598 Å and 1.544426 Å, respectively. The average wavelength of Cu Kα is 1.54278 Å.
|
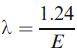 ------------------------------ [4484b]
------------------------------ [4484b]