Chapter/Index: Introduction | A |
B |
C |
D |
E |
F |
G |
H |
I |
J |
K |
L |
M |
N |
O |
P |
Q |
R |
S |
T |
U |
V |
W |
X |
Y |
Z |
Appendix
4f Elements in Periodic Table
| Table 4693a. 4f elements in periodic table.
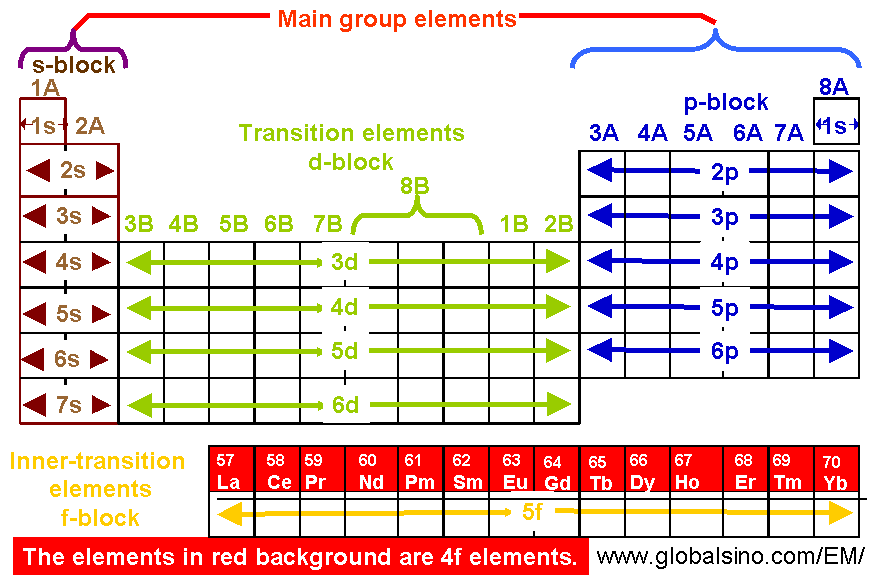
Table 4693b. Electron configurations, filling orbitals, and valence electrons of 4f elements.
| Atomic Number |
Symbol |
Name |
Electron Configuration |
Filling Orbital |
Valence Electrons |
| 57 |
La |
Lanthanum |
1s2 2s2p6 3s2p6d10 4s2p6d10 5s2p6d1 6s2 |
5d1 |
5p16s2 |
| 58 |
Ce |
Cerium |
1s2 2s2p6 3s2p6d10 4s2p6d10f1 5s2p6d1 6s2 |
4f2 |
4f26s2 |
| 59 |
Pr |
Praseodymium |
1s2 2s2p6 3s2p6d10 4s2p6d10f3 5s2p6 6s2 |
4f3 |
4f36s2 |
| 60 |
Nd |
Neodymium |
1s2 2s2p6 3s2p6d10 4s2p6d10f4 5s2p6 6s2 |
4f4 |
4f46s2 |
| 61 |
Pm |
Promethium |
1s2 2s2p6 3s2p6d10 4s2p6d10f5 5s2p6 6s2 |
4f5 |
4f56s2 |
| 62 |
Sm |
Samarium |
1s2 2s2p6 3s2p6d10 4s2p6d10f6 5s2p6 6s2 |
4f6 |
4f66s2 |
| 63 |
Eu |
Europium |
1s2 2s2p6 3s2p6d10 4s2p6d10f7 5s2p6 6s2 |
4f7 |
4f76s2 |
| 64 |
Gd |
Gadolinium |
1s2 2s2p6 3s2p6d10 4s2p6d10f7 5s2p6d1 6s2 |
4f7 |
4f75d16s2 |
| 65 |
Tb |
Terbium |
1s2 2s2p6 3s2p6d10 4s2p6d10f9 5s2p6 6s2 |
4f9 |
4f96s2 |
| 66 |
Dy |
Dysprosium |
1s2 2s2p6 3s2p6d10 4s2p6d10f10 5s2p6 6s2 |
4f10 |
4f106s2 |
| 67 |
Ho |
Holmium |
1s2 2s2p6 3s2p6d10 4s2p6d10f11 5s2p6 6s2 |
4f11 |
4f116s2 |
| 68 |
Er |
Erbium |
1s2 2s2p6 3s2p6d10 4s2p6d10f12 5s2p6 6s2 |
4f12 |
4f126s2 |
| 69 |
Tm |
Thulium |
1s2 2s2p6 3s2p6d10 4s2p6d10f13 5s2p6 6s2 |
4f13 |
4f136s2 |
| 70 |
Yb |
Ytterbium |
1s2 2s2p6 3s2p6d10 4s2p6d10f14 5s2p6 6s2 |
4f14 |
4f146s2 |
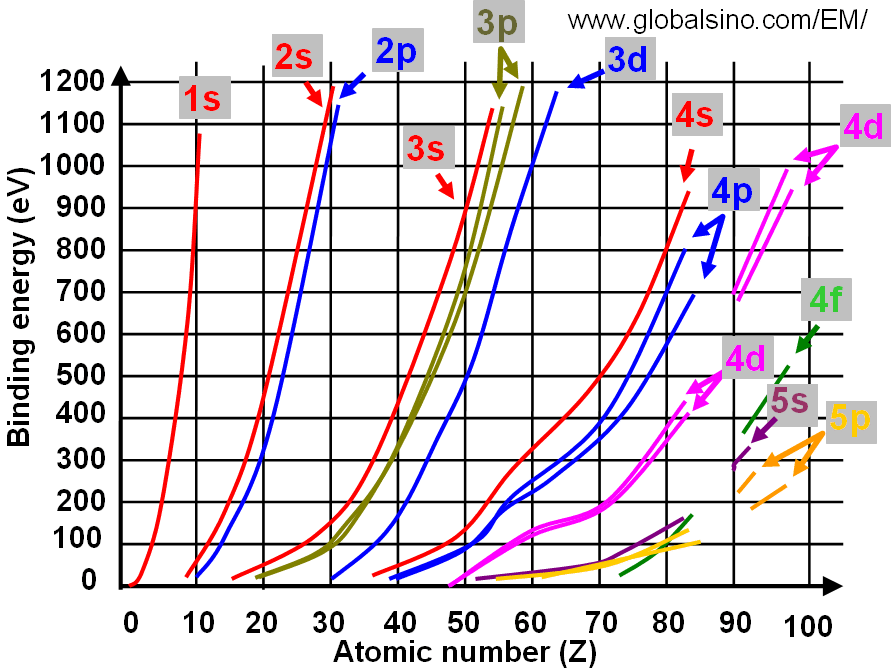
Figure 4693 shows the relation between electron binding energies and atomic number (Z).
Figure 4693. Relation between electron binding energies and atomic number (Z) (at low binding energies).
|